|
Cancer, from the citizens’ perspective, is a life-threatening illness that is more common in elders. It can always be attributed to contaminated living conditions (e.g. air pollution, radiation), inferior living habits (e.g. smoking, drinking alcohol), etc. However, apart from adults, children facing cancer also deserve attention.
There are an average of 1838 children’s cancer cases a year, and around 1 in every 420 boys and approximately 1 in every 490 girls in the UK will be diagnosed with cancer by age 14 as estimated. (Cancer Research UK, 2024). In the writer’s hometown, the number is lower. In Hong Kong, an average of 180 children contract the disease each year – about one child in every 10,000. (Children’s Cancer Foundation, 2024) While breast and lung cancer are most common in adults, leukaemia, brain tumour, and lymphoma are the most prevalent types of cancer children encounter. (Children’s Cancer Foundation, 2024) Since the 1970s, when most children diagnosed with cancer had little hope of survival, cure rates have grown dramatically thanks to the advancement of treatment. As a result, the number of survivors has also significantly increased. (Children’s Cancer Foundation, 2024) The cure rate of childhood cancer has rocketed by 4 times from a mere 20% in the 1960s to 84% nowadays. (Children's Cancer and Leukaemia Group,2024) Treatment of children’s cancer falls under three main categories --- chemotherapy, radiotherapy, and bone marrow transplantation. For chemotherapy, the doctor prescribes a combination of anti-cancer drugs administered orally and by injection. In addition to that, some patients may also require radiation to kill the cancer cells. Bone marrow transplantation is always considered the last line treatment given that finding a match bone marrow is difficult, not to mention the high risk of the operation for such a young child. As a long-term volunteer in a foundation and charity providing support to these children, it is pleasing to see how the improvement in treatment has hugely boosted the cure rates. Nevertheless, the side effects are worrying. They lead to inconvenience to children’s development and lack of self-confidence. Bone fracture has always been a commonplace but concerning side effects of steroids. It restricts the daily physical activities of children. The difference between them and other children may also hurt their self-esteem. Another alarming side effect is the increase in ocular pressure and steroid-induced glaucoma. As symptoms of glaucoma are difficult to define by children, it may already result in severe vision loss when the glaucoma is diagnosed. As pharmacists, we play a pivotal role in the healthcare system, including supporting these children and their families. A cancer diagnosis can sometimes be the first time a parent has had to give their child any medicines, apart from paracetamol. Treatment of paediatric cancer still relies heavily on chemotherapy drugs, whether administered by mouth or via intravenous infusion. (Children's Cancer and Leukaemia Group,2020) The effectiveness of the chemotherapy depends on the medication adherence. We have the responsibility to ensure children receive the medication accurately. We are also responsible for highlighting the possible common mild side effects and alarming symptoms they should stop and find the doctor straightaway. For example, in patients’ counselling, we can recommend lifestyle changes, including supplementation with calcium and vitamin D, diet, and proper exercise. These can slow the rate of bone loss. (Guise TA, 2006) Another way to assist them as a pharmacist is to join the research team. Pharmacists can apply their pharmacological knowledge and be involved in new chemotherapy research and review of international treatment options. This is also my motivation to study pharmacy. These children do need our help and support. They deserve a chance to live their lives. Not only can they grow as other children do, but they can also be the leading icons and professionals in their sector. For example, in Hong Kong, “Outstanding Little Life Warriors Award'' awardee, Yu Chui Yee, is a Paralympics gold medalist in fencing. Many awardees are now doctors and medicine students who aim to contribute to society and help patients. (Little Life Warrior Society, 2019) It is great to see how the advancement in treatment has saved so many lives and we are all looking forward to more children to be benefited. References: Cancer Research UK (2024) ‘Children's cancer statistics’, in Cancer Research UK [Online]. Version. Available at: https://www.cancerresearchuk.org/health-professional/cancer-statistics/childrens-cancers#heading-Three (Assessed: 29 January 2024) Children's Cancer Foundation (2024) ‘Childhood Cancer Facts & Figures’, in Children's Cancer Foundation [Online]. Version. Available at: https://www.ccf.org.hk/en/information/childhood_cancer_facts_and_figures/ (Assessed: 29 January 2024) Children's Cancer and Leukaemia (2024) ‘Survival rates’, in Children's Cancer and Leukaemia [Online]. Version. Available at: https://www.cclg.org.uk/survival-rates (Assessed: 29 January 2024) Children's Cancer and Leukaemia (01 Sep 2020) ‘Back to basics: The pharmacist’s role in treating childhood cancer’, in Children's Cancer and Leukaemia [Online]. Version. Available at: https://www.cclg.org.uk/contact-magazine/the-pharmacists-role-in-treating-childhood-cancer (Assessed: 29 January 2024) Guise TA (2006) ‘Bone loss and fracture risk associated with cancer therapy’, in The oncologist, 11(10), 1121–1131. [Online]. Version. Available at: https://doi.org/10.1634/theoncologist.11-10-1121 (Assessed: 26 January 2024) Little Life Warrior Society (2019) ‘The 4th "Outstanding Little Life Warriors Award" Awardees’, in Little Life Warrior Society [Online]. Version. Available at: https://www.llws.org.hk/pages/hk/Info.aspx?10_42_0_10202_ (Assessed: 29 January 2024) Written by Tim Chan
0 Comments
Pharmacists have a key role to play in society, as they provide a source of information and help to the community. However, by being on the front line, dispensing medicines and treatment for their customers, pharmacists need to be aware of the dangers and risks that arise, and how that may negatively affect their mental health. The mental toll that many healthcare professionals come under is often brushed past, and when it comes to pharmacists, there is no exception. There is an unfortunate rise in pharmacy-related crime in the UK, and with that comes alot of mental strain for the people working behind the counter.
The PDA state how over a third of pharmacists do not feel safe for more than half of their time at work, and 20% don’t feel physically safe at all (2021), and the following year, they conducted an online survey and found that 85% of pharmacists have experienced or work with someone who has experienced verbal or racial abuse that month (2022). A BBC news article even shows how some pharmacists have to carry a rape alarm and dye spraydue to how unsafe they feel from threats (Hayward, C, 2022). The stress and anxiety pharmacists face, and how unsafe they may feel at work have shown to have dire effects for not only the healthcare professionals, but for pharmacies and their customers, too. It can affect the quality of care a pharmacist may be able to provide and can also lead to staff shortages since the PDA concluded that 77% of pharmacists were thinking of changing careers or employment status due to the amount of pressure and mental health issues they face (2023). This would evidently cause many issues with the availability of care for the community. Despite many measures in place to limit dangerous encounters, one being the chief executive of the GPhC making it known that “any abuse of pharmacy staff is completely unacceptable and robust action should be quickly taken in response to any incidents, including by law enforcement” (Duncan Rudkin,2021), these encounters still happen. Most people assume it is a result of substance abusers using pharmacies in the hopes of gaining medicines illegally with prescriptions, yet many other factors come into play. For example, 111 pharmaceutical products are facing supply problems (BMJ 2023;383:p2602), and patients are being told that their medicines aren’t in stock. Difficulties in getting medicines can bring about frustration in customers which can be displaced onto community pharmacists, and staff can be blamed for these shortages. Additionally, waiting time in pharmacies is often long, and patients who have found it hard to access the GP and make an appointment may be further annoyed at the business in community and hospital pharmacies. Even though the pharmacist is not at fault here, customers may feel as though it is, and this can lead to verbal and even physical abuse towards the people behind the counter. The increasing frequency of these disruptions to patient and staff care has led to the creation of the PDA- the Pharmacist’s Defence Association and their copious amounts of resources. For customers, there are posters being displayed in pharmacies that remind them of the zero-tolerance policies towards abuse and violence, and for pharmacists, the PDA has made a “Safer Pharmacies Charter” that outlines how a pharmacist can prioritise their mental health. For example, getting adequate rest, raising concerns and ensuring physical safety are all things a pharmacist can do to feel safer at work . Pharmacies are also taking into account how these events can affect their staff, and are implementing training and therapy. A recent example is a pharmacy in Redditch that started counselling staff after an incident occurred involving a staff member being threatened with a knife (BBC, 2022). Some extreme measures are also being put into practice when appropriate, and pharmacies are asking staff to wear body cameras and installing panic buttons that are linked to police stations. (Isabel Shaw, 2021). Pharmacists are trained to always prioritise patient care, yet many do not realise that in order to optimise the quality of their care, they must feel safe and protected at all times. As pharmacists, self-care should be a major part of your lifestyle, and reaching out for help and resources is always a good idea. As customers, we should always be respectful, considerate, and appreciate that pharmacists are making supreme efforts to provide us with the best of care. References: BBC (2022) Redditch-based pharmacy chain gets counselling for staff over abuse. Available from: Redditch-based pharmacy chain gets counselling for staff over abuse - BBC News Accessed [23/01/24] BMJ (2023;383:p2602) Record number of drugs hit by supply problems, trade body warns. Available from: Record number of drugs hit by supply problems, trade body warns | The BMJ Accessed [23/01/24] Crimestoppers (n.d) Stopping violence in pharmacies. Available from: Stopping violence in pharmacies | Crimestoppers Accessed [23/01/24] GPhC (2021) Abuse or violence against pharmacy staff ‘completely unacceptable. Available from: Abuse or violence against pharmacy staff ‘completely unacceptable’ | General Pharmaceutical Council Accessed [23/01/24] Hayward, C (2022) Community pharmacists speak out against rise in abuse and attacks. BBC, 8 June. Available from: Community pharmacists speak out against rise in abuse and attacks - BBC News Accessed [23/01/24] PDA (2022) PDA highlights ongoing concerns about increasing levels of violence and abuse in community pharmacy. Available from: PDA highlights ongoing concerns about increasing levels of violence and abuse in community pharmacy | The Pharmacists' Defence Association [Accessed 23/01/24] PDA (2022) PDA launches violence and abuse in community pharmacy workplace survey. Available from PDA launches violence and abuse in community pharmacy workplace survey | The Pharmacists' Defence Association Accessed [23/01/24] PDA (2023) Pharmacists echo poor working conditions faced in NHS hospitals. Available from: Pharmacists echo poor working conditions faced in NHS hospitals Accessed [23/01/24] Shaw, I (2021) Pharmacists to start wearing body cameras for protection against patient abuse. The Pharmacist, 3rd December. Available from: Pharmacists to start wearing body cameras for protection against patient abuse Accessed [23/01/24] Written By: Zainab Siddique Figure [1]
Sepsis is a severe medical condition where the body's immune system becomes overly active, leading to potential life-threatening complications such as shock, organ failure, and death. It can affect individuals across age groups and health statuses, although it is more prevalent in very young children, older adults, and those with pre-existing health conditions. The reasons for the development of sepsis in response to common infections remain unclear, making early recognition and prompt treatment crucial for preventing severe consequences. This essay delves into the pharmaceutical perspective on sepsis in the context of the UK, navigating the role of pharmacy in septic early diagnosis and exploring challenges and innovations in treatment. Within the UK, sepsis poses a significant challenge to the NHS, demanding timely and effective interventions. The unique aspects of the UK healthcare system, including the centralised NHS structure, accentuate the importance of early detection and swift intervention to improve patient outcomes. In particular, monitoring vital signs in a pharmacy allows pharmacists to contribute to the early diagnosis of sepsis. Hence, pharmacists can play a proactive role in identifying potential sepsis cases and promptly referring patients for further medical evaluation by closely observing these crucial physiological parameters. [2] The effective treatment of sepsis is hindered by the rise of antimicrobial resistance (AMR). AMR occurs when specific strains of bacteria, viruses, fungi, or parasites adapt to evade the effects of antimicrobials, including antibiotics, antivirals, antifungals, and antiparasitics designed to combat them. As more pathogens become resistant to these drugs used for infections, the risk of sepsis increases. This poses a significant challenge in the UK healthcare system, leading pharmaceutical research to explore innovative antibiotics. Collaborative initiatives with the NHS focus on implementing antimicrobial stewardship programs. The successful management of sepsis is intricately linked to the critical battle against antimicrobial resistance. [3] While previous sepsis drug candidates failed in clinical efficacy, recent advancements in understanding sepsis pathways have led to the development of promising drugs. Rather than a singular sepsis profile, various phenotypes have been identified, allowing for targeted drug development. New candidates focus on Pathogen-Associated Molecular Patterns, the Coagulation System, the Endothelium, End-Organ Damage, and Host Immune Response. These drugs, tested in randomised controlled trials, aim to reduce organ dysfunction and improve patient outcomes. The evolving understanding of sepsis phenotypes, biomarkers, and intervention timing suggests a more personalised and effective approach, similar to current cancer therapies, may emerge in the near future. [4] In summary, sepsis poses a formidable challenge in the UK's healthcare system, requiring a collaborative effort from healthcare professionals and pharmaceutical research. Pharmacists play a crucial role in early sepsis diagnosis by monitoring vital signs. The escalating concern of antimicrobial resistance underscores the need for joint initiatives between pharmaceutical researchers and the NHS to develop innovative antibiotics. Recent breakthroughs in understanding sepsis phenotypes offer hope for more personalised treatment approaches, resembling strategies in cancer therapies. The ongoing collaboration between healthcare professionals and researchers is essential for advancing sepsis management and improving patient care. References 1. Comanche County Memorial Hospital Community (2023) ‘Sepsis: A Silent Killer’ in Community Health [Online]. Available at: https://www.ccmhhealth.com/sepsis-a-silent-killer/ (Accessed on 8 January 2024) 2. United Kingdom Sepsis Trust (2022) ‘Yellow Manual 6th Edition’ in The UK Sepsis Trust [Online]. Available at: https://sepsistrust.org/wp-content/uploads/2022/06/Yellow-Manual-6th-Edition.pdf (Accessed on 8 January 2024) 3. Sepsis Alliance (2022) ‘POWER the AMRevolution’ in Antimicrobial Resistance [Online]. Available at: https://www.sepsis.org/power-the-amrevolution/ (Accessed on 8 January 2024) 4. Vignon, P., Laterre, PF., Daix, T. et al. (2020) ‘New Agents in Development for Sepsis: Any Reason for Hope?’ in Drugs 80 [Online]. Available at: https://doi.org/10.1007/s40265-020-01402-z (Accessed on 8 January 2024) Written by: Dasom Lee Ethical considerations often form the backbone of pharmacists' responsibilities, and pharmacy professionals have to find the delicate balance between dispensing medications and upholding patient autonomy. In the field of pharmacy practice, ethical considerations are the foundation on which pharmacists build their responsibilities. This blog focuses on major ethical dilemmas faced by pharmacy professionals in the UK, highlighting practical scenarios and potential solutions.
One of the most frequent ethical dilemmas that pharmacists face is encountering patients who deviate from prescribed instructions. The balance between persuading patients to adhere and respecting their autonomy is significant, especially when noncompliance risks significant side effects. With over 50% of patients facing this issue, pharmacy professionals navigate the fine line between advocacy and patient preference. Also, challenges arise when patients request quantities exceeding prescribed limits, whether due to severe pain or special circumstances. While accommodating such requests may be tempting, strict legal limits exist. Making a rational decision while empathising with genuine pain and regulatory adherence is pivotal. Exceeding medication limits could result in severe consequences, including licence loss. Maintaining impartiality is essential for pharmacy professionals as well. Guarding against personal biases ensures consistent interactions with patients. Refusing to dispense prescriptions based on personal beliefs, such as refusing to dispense prescribed birth control from a qualified prescriber, is unethical. A neutral stance is paramount, acknowledging the significant role pharmacy professionals play in ensuring unbiased care for every patient regardless of their religion, age, gender, race, and other personal attributes. This study, conducted in Kerman in 2018, is worth paying attention to. This investigation aimed to identify the challenges of ethical behaviors in the drug supply domain within pharmacies using a principles-based approach. The research focused on the ethical principles of autonomy, beneficence, non-maleficence, and justice, guided by the "Beauchamp and Childress Ethics" theory. The qualitative data analysis included interviews with pharmacy technicians, patients, pharmacists, insurance company inspectors, and other professionals in the pharmaceutical industry. The findings, derived from the analysis of data gathered through interviews, revealed eight main categories and 26 subcategories of ethical challenges in the drug supply at pharmacies. The primary categories included patient privacy, patient independence, communication principles, patient-centered services, drug suppliers, patient harm avoidance, supervision, and distributive, procedural, and interactional justice. Notable subcategories encompassed increasing patient awareness, culturizing prescription, rational drug use, confidentiality, and privacy. In conclusion, the study highlighted that the most significant challenges in ethical behaviors in the pharmacy domain were related to the principles of autonomy and beneficence. The implications of these findings suggest a need for healthcare to emphasize patient independence, privacy, and patient-centered services. The study's results can serve as a valuable resource for policymakers as well, aiding in the development of educational content, the establishment of professional ethics charts in pharmacies, and the implementation of accreditation measures to address these ethical challenges effectively. Pharmacy ethics in the UK are a powerful force shaping healthcare delivery. Pharmacists, armed with knowledge, compassion, and ethical commitment, navigate complex responsibilities to ensure each prescription reflects a dedication to patient care. Reflecting on these ethical considerations underscores the indispensable role pharmacists play in upholding the moral integrity of healthcare, where the pursuit of well-being is guided by principles grounded in compassion and professional responsibility. References: Iranmanesh, M., Yazdi-Feyzabadi, V. & Mehrolhassani, M.H. The challenges of ethical behaviors for drug supply in pharmacies in Iran by a principle-based approach. BMC Med Ethics 21, 84 (2020). https://doi.org/10.1186/s12910-020-00531-0 Written by: Seungeun Cho Figure 1 [3] Geriatric bloodwork analysis stands as a fundamental component within the realm of pharmaceutical practice, especially when considering the various ailments that can happen concurrently in the elderly. During Term 1 that has just passed, I had the privilege of attending a hospital placement experience within the Care of Older People (COOP; or geriatrics) team at an NHS trust located in North London as part of a module coursework assignment. This experience offered invaluable insight into the critical role that pharmacists play in healthcare, especially in the senior population. Throughout this experience, I got to live life as a hospital pharmacist (albeit only for 2 weeks); screening prescriptions, looking for drug interactions, and establishing drug histories for newly admitted patients as part of the medicines reconciliation process. However, it is through performing these seemingly routine tasks where I was able to dive into the mindset of a hospital pharmacist, who places their patients’ healthcare as their priority. It is also during this placement that I recognised the importance of being detail-oriented, as minor alterations in blood tests can signal substantial underlying issues which demand a pharmacist’s meticulous attention. Unlike other specializations, COOP underscores the significance of polypharmacy, as many patients often have various ailments that require medical intervention simultaneously. For instance, an elderly patient with cancer may also have hypertension, hence it is not alarming to see cyclophosphamide on their drug chart with amlodipine right beneath it. Therefore, as a pharmacist, it is our job to ensure that all the drugs on the patient’s drug chart are safe, ie:
However, without providing any additional context, one might assume that a pharmacist would just sit at a patient’s bedside 24/7, eyes peeled and stuck onto the patient - because surely, that’s what monitoring means, right? Well, this is where the essence of this entry comes into play. Blood tests, or referred to bloodwork hereinafter, are essentially how pharmacists glue their eyes on their patient to pick up on any abnormalities, or signs that they need to stop or start a certain medication. Comprehending geriatric blood investigations involves the meticulous consideration of many biological markers, including haemoglobin count (Hb), red and white blood cell counts, platelet levels, coagulation factors such as INR, inflammatory markers such as CRP levels, as well as electrolyte concentrations such as sodium, potassium, and bicarbonate; to name a few. However, with that being said, the true challenge lies not merely in identifying these components, but rather the interpretation of their implications within the broader context of pharmaceutical care. This understanding of the bloodwork components must also come with fingertip clinical knowledge that is expected from the pharmacist, in order to produce the most effective patient care strategy. For example, a 79-year-old patient admitted for a chest infection, has been taking frusemide for the past 12 years, is said to have acute kidney injury (AKI). By looking at the patient’s bloodwork, their serum creatinine levels to be exact, will we be able to obtain a value to perform their creatinine clearance calculation, which is indicative of kidney function. Therefore, with this in mind, we will be determining if frusemide, the loop diuretic, is useful for them. Knowing that frusemide is cleared renally [2], the drug poses more harm for the patient than good, as processing the drug would burden the patient’s kidneys further and worsen their AKI. Therefore, this discussion can be brought up to the other healthcare professionals looking after the patient, to decide if frusemide can be temporarily taken off their drug chart until their creatinine clearance improves. Other than that, medications used for the patient’s presenting complaint (chest infection), should also be screened by the pharmacist to ensure that the patient will get treatment from their presenting complaint, whilst not worsening any of their already existing ailments. Therefore, by honing our skills in understanding and analysing these investigations, coupled with the clinical knowledge that the school is bequeathing to us, we equip ourselves with the tools necessary to provide informed and empathetic care to our senior patients. As we decode these results, it is imperative that we, as pharmacists, keep in mind that these aren’t just figures on a screen. These are our patients’ stories, and their cries for help. Every decision that we make has the potential to change not only our patients’ lives, but the lives of those they love as well. Let’s keep decoding and making a difference in geriatric care - one blood report at a time. References: 1. MedicinesComplete — BNF | Hyoscine Butylbromide | Contraindications [Internet]. www.medicinescomplete.com. 2023 [cited 2023 Dec 25]. Available from: https://www.medicinescomplete.com/#/content/bnf/_881463863?hspl=hyoscine%20butylbromide#content%2Fbnf%2F_881463863%23pot-contraindications 2. DrugBank Online. Furosemide [Internet]. go.drugbank.com. 2023 [cited 2023 Dec 25]. Available from: https://go.drugbank.com/drugs/DB00695 3. Sabah D. 10 tips for a healthy old age [Internet]. Daily Sabah. 2019 [cited 2023 Dec 31]. Available from: https://www.dailysabah.com/health/2019/04/22/10-tips-for-a-healthy-old-age Written by: Olivia Ho 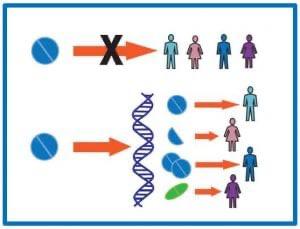 Pharmacogenomics, a fusion of pharmacy and genomics, is revolutionizing healthcare by leveraging genetic information to tailor medication treatments to individuals. This field is a cornerstone of personalized medicine, an approach that considers unique genetic, environmental, and lifestyle factors in disease treatment and prevention. As the medical world increasingly recognizes that 'one-size-fits-all' doesn't apply to medication efficacy and safety, pharmacogenomics is gaining unprecedented importance. Pharmacogenomics lies in understanding how genetic variances affect drug metabolism and efficacy. Individuals metabolize drugs differently, primarily due to genetic differences in enzymes responsible for drug metabolism. These variations can significantly impact the effectiveness and safety of medications, leading to varying therapeutic outcomes among patients. A striking example is the CYP2D6 gene, which plays a vital role in metabolizing antidepressants. Variations in this gene can lead to drastically different responses to the same medication. For instance, some individuals with certain CYP2D6 variations may metabolize drugs too quickly, reducing drug efficacy, while others may metabolize too slowly, increasing the risk of side effects. Healthcare providers now increasingly rely on pharmacogenomic testing to guide medication selection and dosage. This testing identifies specific genetic markers that predict how a patient will respond to a drug. With this knowledge, providers can tailor treatment plans, avoiding ineffective medications and reducing the risk of adverse drug reactions. Despite its potential, pharmacogenomics faces challenges, particularly regarding ethical considerations. Issues such as genetic data privacy, potential discrimination based on genetic information, and accessibility of pharmacogenomic testing are critical concerns that need addressing to ensure equitable and ethical implementation. For patients, understanding their pharmacogenomic profile can be immensely beneficial. It empowers them with knowledge about how their body might react to certain medications, allowing for more informed healthcare decisions and potentially leading to better treatment outcomes. The future of pharmacogenomics holds promise for more targeted drug development. As our understanding of genetic influences on drug response deepens, we can expect the emergence of new medications designed for individuals with specific genetic profiles, heralding a new era of truly personalized medicine. Pharmacogenomics is not just a scientific advancement; it's a paradigm shift in healthcare. By aligning genetic insights with clinical practice, it paves the way for more effective, safer, and personalized treatments. Its integration into mainstream medicine, despite challenges, signifies a significant step towards a future where healthcare is tailored to each individual's genetic blueprint. References : Roden, D.M. and Tyndale, R.F., 2013. Pharmacogenomics. The Lancet, 362(9390), pp.1619-1628. Weinshilboum, R. and Wang, L., 2014. Pharmacogenomics: Precision medicine and drug response. Mayo Clinic Proceedings, 89(5), pp.609-621. Johnson, J.A., 2013. Pharmacogenetics in clinical practice: How far have we come and where are we going? Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 33(6), pp.612-625. Written by Cecilia Perdikidi Transitioning to University Life at UCL SOP : Thoughts of Local and International Students11/29/2023 Starting anything new can be very daunting- especially when you are very passionate about it. When I first came to UCL’s School of Pharmacy, I had many mixed feelings- fear, excitement, anxiety. However, these past few weeks have allowed me to settle in well, and I am keen to continue my journey as a pharmacy student. As a Londoner, I had one big advantage- I was familiar with many aspects of the UK, such as the area, the culture and the people. This was very useful when applying my knowledge to pharmacy in the UK.
Living here my whole life meant I had a good grasp of what the rules and regulations were like here. For example, I could tell which medicines were GSL products and which were PoM based on what was available over the counter at Boots. I have also been aware of pack restrictions on paracetamol, so learning about it in my course allowed me to link my experience to my knowledge of medicines. I am also very fluent in what prescriptions are like here, as I always used to glance over them whenever my parents brought them home from the GP. I have seen how the dispensing system works, and what it is like to be a patient picking up a prescription. I now get to see what pharmacists do behind the counter in the UK, and so I get to grasp a fuller picture. The NHS is one of the unique characteristics of the UK. I have been exposed to how overcrowded GPs and hospitals can be, due to the free healthcare provided, and in a way, it has allowed me to understand how much healthcare professionals are needed by the community. Having family friends here who work for the NHS has also proved this to me, as the stories and experiences they share are incredibly inspiring. I am fortunate to benefit from free healthcare, and I hope many more people will be too. Brexit was, to say the least, a huge change for the UK. I witnessed how all areas of the healthcare system were affected, and I was very interested to learn more about how pharmacies specifically had to adapt to the UK leaving the EU. I believe that if I didn't live here, I wouldn't know as much about these changes and so I am grateful that I have a good insight into it. Overall, I have a vivid understanding of pharmacy in the UK, yet there is a lot to learn. Although commuting can be tiring, and I am less aware of what pharmacy looks like globally, I am looking forward to implementing my vast knowledge of being a UK citizen into my degree, and eventually being able to help my community out as a pharmacist. Written by Zainab Siddique, Local First Year Student at UCL School of Pharmacy ----------------------------------------------------------------------------------------------------------------------------------------- Studying abroad can be extremely daunting for an international student. I felt like I was forced to grow up overnight, when I had everything I owned packed into 4 large suitcases and hop onto a plane - all while knowing my heart was beating 10,000km away from my body. However, I still appreciate this opportunity of a lifetime, as these hurdles, though significant, have been integral to my growth and understanding of this new landscape. Cultural adjustment hit hard upon arrival. The expectation to get used to a new culture, new environment, and new people brought about a wave of culture shock, leading to bouts of homesickness. Having to adapt to unfamiliar customs and social norms can undoubtedly be a steep learning curve to most international students. Besides, language barriers are evident to be a formidable obstacle for plenty of international students. While I was fortunate enough to not have encountered this hurdle personally, I know of many others who struggled with understanding intricate academic and medical terminologies - just because their first language wasn’t English. The struggle to communicate and comprehend effectively proved to be a constant obstacle for this particular group of students, undoubtedly requiring them to put in additional effort to bridge the linguistic gap in order to survive. Visa and immigration - definitely not the most thrilling subjects, but undeniably crucial. Most international students had to learn how to manage these arduous tasks without any guidance. Like stepping on a tightrope, I personally felt overwhelmed when it came to this part of moving abroad, as I knew that this was of paramount importance when it came to securing part-time work opportunities that were essential for financial stability. Speaking of finances - the topic weighs even more heavily on international students, as we have to think about tuition fees and the soaring cost of living - all while worrying about the unpredictable currency exchange rates. Though a life skill that is essential for all to learn, international students had to prioritise thorough budgeting. Some might even opt to seek scholarships, which is another burden to add onto a pair of heavy shoulders. Lastly, to top everything off, the ever-unpredictable London weather! Prior to moving here, I never knew that the change from a radiant sunny afternoon to a gloomy, rainy day can happen within minutes. Therefore, adapting to this unpredictable climate was a daily adventure in itself. International students who come from tropical countries (like myself) would also find difficulty in getting accustomed to the cold weather especially during the winter season. However, each of the above challenges has served as a defining chapter in my journey as a student at the UCL School of Pharmacy. I have learned to be a resilient individual that is able to change the sails on my ship according to the winds, achieving invaluable lessons in perseverance amidst adversity. Written by Olivia Ho, Third Year Student at UCL School of Pharmacy from Malaysia ---------------------------------------------------------------------------------------------------------------------------------------- As a Korean international pharmacy student at UCL Pharmacy School, my academic journey has been faced with both personal challenges and a basic understanding of South Korea's healthcare system. One of the challenges has been struggling with pharmacy concepts in a foreign language, English. This hurdle has required a dedicated effort to bridge the language gap and adapt to a new academic environment. However, through perseverance and active engagement, I think I can enhance my language proficiency and academic skills simultaneously. Turning the focus to South Korea's healthcare system, it is designed to give all residents accessible and essential healthcare. The National Health Insurance (NHI) program is crucial, covering both Korean citizens and foreign residents. It's mandatory for everyone, with coverage based on income and employment, ensuring fair access to healthcare resources. What’s interesting is that private insurance is prevalent in South Korea, offering additional benefits such as dental coverage, operations coverage, and more, to complement the NHI program. In Korea, the healthcare system is organised into primary, secondary, and tertiary care; with local clinics providing primary care, general hospitals offering secondary care, and specialised medical centres delivering tertiary care respectively. Also, preventive healthcare is vital through regular health check-ups and screening programs for employees. With various care systems, there are various healthcare professionals in Korea including traditional Korean medicine practitioners, contributing to a robust healthcare ecosystem. This diverse array of professionals ensures that individuals receive holistic care, addressing both conventional and traditional aspects of health. One of the healthcare sectors, pharmacy in Korea, operating hours reflect the commitment to accessible healthcare; with 24-hour pharmacies in large cities and standard hours from 9 am to 6-7 pm on weekdays and Saturdays. Furthermore, some Korean pharmacies integrate traditional Korean medicine with Western medicine, making herbal remedies and oriental medicine products readily available to the public. Looking back on my time as an international pharmacy student and exploring South Korean healthcare has been valuable. Overcoming language barriers and adapting to a new academic setting has been transformative. This experience has widened my view on global healthcare and sparked an interest in understanding healthcare in different countries. It also prompts me to consider cultural awareness and sensitivity. These insights will play a significant role in my future as a pharmacist, as I aim to promote a comprehensive and inclusive approach to patient care. Written by Dasom Lee, First Year Student at UCL School of Pharmacy from South Korea ----------------------------------------------------------------------------------------------------------------------------------------- Being able to study abroad in such a great pharmacy school has always been my pleasure. Since I received the offer, I have been looking forward to new cultures, food, and friends I may be in touch with. It allows me to broaden my horizons. However, what I did not expect was the mixed feelings I had to encounter. As soon as I stepped into the Hong Kong International Airport, my feelings altered quickly. I pretended to be so relaxed, but I was worried about my new life, and the cultural difference I may face. Without direct support from friends and family, I was forced to grow up, live independently, and self-encourage when facing obstacles. Being homesick is inevitable for an international student, and I am so glad I have built cohesive friendships in my hometown, and they are always there to support me. Despite the challenges, I still feel very grateful to study here, and allow me to jump out of my comfort zone. Speaking of the course I am taking --- Pharmacy, the role of pharmacists is quite different in these two places because of the difference between their healthcare systems. In Hong Kong, the healthcare system mainly consists of hospitals and clinics. Public hospitals are funded by the government and administered by the Hospital Authority, a department like the NHS, while private hospitals are self-funded. There are also private clinics around the city that allow citizens to seek treatment for some basic illnesses. Doctors are responsible for prescribing and dispensing medicines as Hong Kong does not adopt the system of separation of prescribing and dispensing. Citizens mostly visit the A&E or private clinics when they are not feeling well and seldom, if not never look for a community pharmacy. The pharmacists in Hong Kong can be divided into hospital and community pharmacists. They have no power to prescribe medicines. Community pharmacists usually provide basic healthcare consultation and dispense the prescribed medicines. While for hospital pharmacists, apart from conducting the job community pharmacists do in the pharmacy, they may also join the medical team in providing professional comments on the dose and choice of medicines for patients. I am glad to share with you all my experiences and feelings so far to study here as an international student and to explain more about the healthcare system in Hong Kong. Written by Tim Chan, First Year Student at UCL School of Pharmacy from Hong Kong -------------------------------------------------------------------------------------------------------------------------------------------- Embarking on the journey of moving from Greece to the UK and specifically to London to study pharmacy at UCL , I found myself having a mixed of emotions and facing a lot of challenges . It was a plunge into the unknown, a journey through diverse landscapes, both personal and professional. The awareness of the difficulties that might lay ahead acted as a guide for mental preparation. The vibrancy of London epitomised by the intricate tube map and sprawling distances, initially struck fear into my heart. Yet, each challenge became an opportunity for growth . Acceptance into UCL was a dream come true. I was happy about the outcome but nervous about the competitiveness of the course. One of the highlights of this journey has been the excitement of meeting new people from all corners of the globe. London is a melting pot, and UCL mirrors that diversity. The prospect of learning and growing alongside individuals from diverse backgrounds has been a driving force that keeps me enthusiastic about each day. Since it was my first time living abroad, away from family , the responsibilities piled up, from signing up for a GP to opening a bank account, getting a new UK phone number, and finding affordable accommodation in central London. The pressure was on, and the learning curve was steep. It felt like adulting on fast-forward. Regarding healthcare in Greece , we have the National Healthcare Service (ESY), a lifeline for most of us. It's funded by contributions from employees, employers, and government subsidies. Waiting times for non-emergencies in the public sector can be a bit much. From a pharmaceutical aspect , in my studies and through my placements I realised that in the UK , pharmacies offer services like flu vaccinations and medication reviews , something I didn't see much of back home.In Greece however , pharmacies could be like social hubs, especially in quieter areas. Casual conversations especially from the elderly to the pharmacists as well as advice flow freely, creating a unique sense of community. Some medications might need a prescription, but Greek pharmacists might be able to dispense just certain drugs that are not over the counter without one . As I sit here reflecting on my journey so far , I can't help but smile . It has been a series of challenges , but each one has been a stepping stone to personal growth . From navigating the chaos of London to immersing myself to the fascinating world of pharmacy . It's an adventure I wouldn't trade for anything . Here is to embracing the unknown , stepping out of comfort zones and finding beauty in the journey. Written by Cecilia Perdikidi, Second Year Student at UCL School of Pharmacy from Greece -------------------------------------------------------------------------------------------------------------------------------------------- Being an MPharm student at UCL School of Pharmacy is an exciting journey filled with academic exploration, cultural adjustments, and the pursuit of becoming a registered pharmacist in the highest quality institution. Throughout this, I encountered unique challenges which I struggled with at the beginning of the year but discovered a newfound appreciation for life in both the UK and my home country, South Korea. The UK's education system, especially at universities like UCL, emphasizes independent study and research, offering academic freedom. As an international student, this means taking on the responsibility of navigating coursework independently, which can be time-consuming and costly, considering expenses like flights and housing. Nonetheless, the exposure to UCL's diverse and dynamic learning environment is an invaluable experience that goes beyond academics. In London, the consistently mild weather and language barriers sometimes create social challenges, leading to moments of homesickness and even depression. I actually struggled during my last year in the School of Pharmacy, especially when I felt overwhelmed with a significant amount of work to do. In contrast, South Korea's four distinct seasons and warm culture provide a comforting backdrop, making it easier for those who find solace in the familiarity of changing climates. South Korea has become a global healthcare success story, with the National Health Insurance Service (NHIS) offering comprehensive coverage and ensuring accessibility for all. The network of hospitals, along with affordable medical services, eliminates the majority of concerns about health insurance or the cost of medical care. Visiting a doctor(GP) or getting a prescription in South Korea is straightforward and efficient, fostering a sense of comfort and relaxation when dealing with health issues. While the UK's NHS is renowned for providing medical services regardless of financial status, it's crucial to recognize that South Korea's healthcare system brings advantages regards efficacy and accessibility. The systematic nature of South Korea's healthcare services may pose challenges, but the overall experience is marked by a sense of ease and reassurance. South Korea's healthcare success serves as a reminder of the diverse strengths different parts of the world contribute to ensuring the well-being of their populations. However, being an MPharm student at UCL School of Pharmacy involves a multifaceted journey of academic growth, cultural adaptation, and the pursuit of excellence. Despite the challenges, the experience is enriched by the global perspective gained from studying in central London. Written by Seungeun Cho, Second Year Student at UCL School of Pharmacy from South Korea References: Arrival Guides (2021) ‘Pharmacy - Seoul’ in Arrival Guides [Online] Available at: https://www.arrivalguides.com/en/Travelguide/SEOUL/essentialinformation/pharmacy-24982 (Accessed on 27 November 2023) National Health Insurance Service (2020) ‘Health Security System’ in National Health Insurance [Online]. Available at: https://www.nhis.or.kr/english/wbheaa02300m01.do (Accessed on 27 November 2023) Real Reporting Foundation (2021) ‘SOUTH KOREA: HEALTHCARE WORKERS’ in World Health Systems Facts [Online]. Available at: https://healthsystemsfacts.org/national-health-systems/national-health-insurance/south-korea/south-korea-health-system-personnel/ (Accessed on 27 November 2023) I recently had my early community immersion experience this past January and February in Asheville, NC. During my time, I kept hearing about an Adderall shortage, and would answer phone calls daily from patients wondering if more Adderall had come in. I was curious as to how there was a shortage, as I couldn’t really imagine that manufacturers simply couldn’t make enough of the product. After doing some research, I found a helpful article written by Matt Stoller, that explains how the shortage is a result of monopolies and has origins in the opioid crisis.
I’ll summarize some of the things I learned and link Matt Stoller’s article below for more in-depth information, if you’re interested. In short, the opioid crisis was the product of “absentee ownership”. Big wholesalers like Cardinal Health, McKesson, and Amerisource Bergen distributed medications neglectfully, oftentimes to pharmacies that were fronts for organized crime. These massive wholesalers weren’t doing this maliciously, or even to try and sneak a profit. They were simply too large to know their clientele, and just didn’t pay attention. As a result, controlled substances made their way to the streets and brought about the opioid crisis. Eventually, there was a settlement where distributors paid money to resolve state litigation involving opioids, for the sake of justice and expelling illicit profits. In addition to this payment, distributors had to follow new rules that rationed the controlled substances they could sell. They had to monitor for “red flags” amongst their customers, a big one being that if the amount of a controlled substance a customer dispenses has grown too much, then they can be “blacklisted” and the wholesaler will not sell them anymore controlled substances. The issue is that wholesalers use algorithms to calculate the quantities their customers can sell. Since the settlement, it has become increasingly difficult for pharmacies to explain to their wholesalers if they have an increase in demand, as the caps set by the algorithm are incredibly rigid. However, this results in a vicious cycle. If one pharmacy hits their algorithmic cap and is blacklisted, then patients have to get their controlled medication from a different pharmacy. If these customers all go to a different pharmacy, then eventually that pharmacy will reach its wholesaler’s cap, and the situation just continues to worsen. It is technically possible for pharmacies to just get their controlled substances from smaller wholesalers that don’t have these algorithms. However, pharmacies are locked into contracts with big wholesalers usually to purchase at least 90% of their generics from them. They might be able to get some controlled substances from these smaller wholesalers, but not nearly enough to meet demand. So, there’s a deadlock – pharmacies can’t get their controlled substances from big wholesalers after they’ve reached the cap, and they also can’t get enough from smaller wholesalers because they’ve already signed a contract to purchase a massive majority of their medications from the big wholesaler. As a result of the opioid crisis, there is an Adderall shortage, community pharmacies suffer from absentee ownership from their wholesalers, and smaller wholesalers are unable to get into the market. If you’re interested in learning more, I again encourage you to check out what Matt Stoller has written and discovered about the topic! Written by: Andrew Nguyen Source: https://mattstoller.substack.com/p/the-monopolies-behind-the-adderall?utm_source=substack&utm_medium=email Why Women's Health is Important
Women’s health is a broad area in pharmacy. According to America's Health ranking, there were 28.5% unintended pregnancies in the U.S in 2022. About 95% of unintended pregnancies occurred because of not using contraception or incorrect use of contraception (1). What is a Pharmacist's Role in Women’s Health It is important to have pharmacists to help women because they are often the first point of contact for women seeking healthcare advice. Pharmacists are able to tell them how to use contraception correctly and what other options, besides traditional condoms, they have to protect themselves from getting pregnant. With help from pharmacists, women can have their own personalized care based on their preferences. Besides physical protection, medications such as birth control pills and Plan B are also available. However, these medications require pharmacy counseling to ensure that women fully understand how they work. There are often myths about birth control medications: “Birth control will make me gain weight”, “birth control will cause cancer”, “plan B is an abortion pill”, “birth control will affect my ability to have children in the future”.... (2). These myths have prevented some women from trying these medications and caused them to lose protections they could have. Pharmacists are important to clarify and guide women to take birth control medications correctly. Besides birth control, pharmacists are also important in supporting and helping patients to treat conditions that are specific to women, such as menstrual cramps, urinary tract infections, and pregnancy related issues. There are certain medications prohibited from use during breastfeeding as well that pharmacists can advise on. Future Opportunities Nowadays, hospitals have opened more specific departments in women's health areas such as women’s nutrition, hair loss, and genital health. Pharmacy is keeping up with this, as well. In the future, pharmacists may be able to focus more on these specialties and provide more personalized care for women. This will also allow women to have greater access to health care services. Written by: Crystal Wang References:
|
�
Categories
All
Archives
October 2022
|